How Do You Know If Its Linear Or Bent?
 Caleb Butler
Caleb Butler 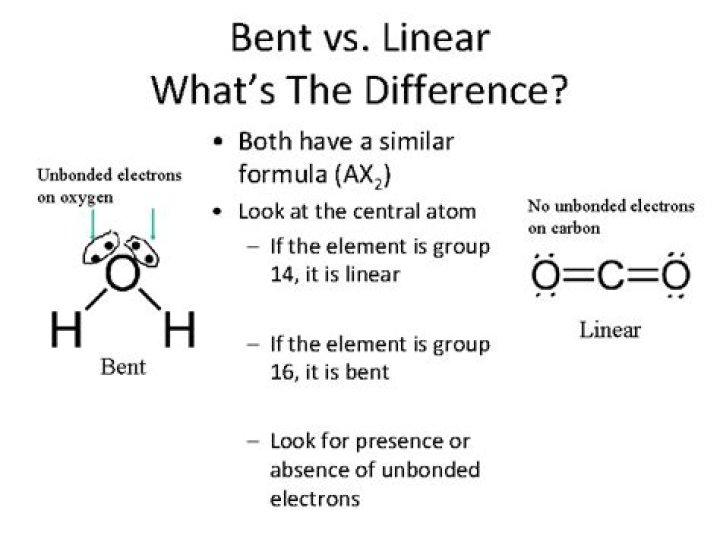
If you look at the structure of linear and bent molecules side by side, you’ll notice that linear molecules have atoms that are tightly bound together, while bent molecules have the opposite arrangement.
Silky Terrier Dog Breed Playing Aro... Silky Terrier Dog Breed Playing AroundHow do you know if something is bent or linear?
A 180°-angled line of atoms is what linear = refers to. The total number of atoms is 2, possibly 3. The greater the number of Lone Pairs, the greater the bend and the smaller the degree. Bent = Linear but bent.
How do you determine if a molecule is linear or nonlinear?
Molecules can be classified as linear or nonlinear depending on whether or not their chemical structure is in the form of a straight line, zigzag, or cross-linked. Acetylene, carbon dioxide, hydrogen cyanide, and a host of other chemicals are examples of reactive gases that can be used in explosives.
What does it mean if a molecule is bent?
Bending molecular geometry, or angular or V-shaped, is a term used in chemistry to describe molecules that have two adjacent bonds that are not coterminous. When two or more covalent bonds are formed between atoms that have different electron configurations, they will almost always form in non-collinear directions.
What is bent example?
There is a difference between a person who is bent on doing something and someone who isn’t. When someone has their heart set on attending a particular university, that person is said to be bent on it. adjective. To say something is “bent” is to say it is not in a straight line. A candy cane is an example of something bent.
What is the shape of linear?
Linear molecules have atoms arranged in a straight line (less than a 180° angle) throughout their structure. The central atom of molecules with linear electron pair geometries is sp-hybridized.
How do you find the linear structure?
Bonded pairs are the number of electrons attached to the central element of a binary compound by the number of substrates attached to the central element of the binary compound. AX2 = Linear Geometry for the BeCl2 structure with #BPrs = 2. Thus, the molecule is defined as being linear by the formula: Cl-Be-Cl.
What is the bent angle?
CONSIDERED (ANGULAR or V-SHAPED)The molecule is bent as a result of the electron repulsion caused by the extra electrons. If two pairs of nonbonding electrons are present, the angle of a bent molecule is less than 109.5 degrees, which is less than 120 degrees.
Is h2o bent or linear?
Heterohedral electron pair geometry results in H2O’s bent molecular shape. As a BENT molecule, the water molecule is so well-known that remembering that it is a BENT molecule is an excellent idea.
What makes a molecule linear?
Atoms in a linear molecule are aligned at an angle less than 180 degrees. In molecules with linear electron-pair geometries, the sp hybridization occurs at the central atom. Examples of a linear electron pair and molecular geometry include carbon dioxide (O=C=O) and beryllium hydride (BeH2).
What defines a linear molecule?
Linear molecules have atoms arranged in a straight line (less than a 180° angle) throughout their structure. The central atom of molecules with linear electron pair geometries is sp-hybridized. Carbon dioxide (O=C=O) and beryllium hydride (BeH2) are examples of linear electron pairs and molecular geometry.



